March 20, 2026
Thank you for all your efforts on this study so far!
Please join our medical team for a drop in discussion, open to all sites and team members to discuss challenges, provide feedback or discuss medical execution of the VANQUISH-1 & 2 protocols. We hope to see you there!
Below are the available dates and times
Please see the updated study supply request form previously sent via email. To ensure your request is actioned promptly please ensure to email completed forms to L.Sheffield@Medpace.com, E.adamson@Medpace.com, S.Steketee@Medpace.com, and S.Gordon@medpace.com.
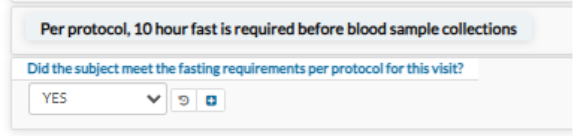
To ensure your malnourishment assessment samples can be tested and resulted, please reference the QRC/Lab Manual guidance for collection and procession of samples, specifically minimum sample volumes and shipment conditions.
Please note:
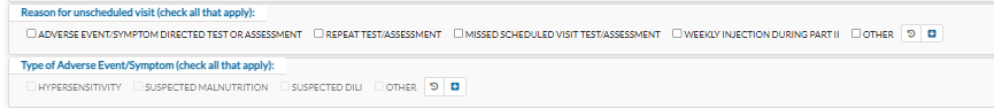
EDC has multiple updates which will require updated data entry on some previous forms. If any of the forms were previously completed for a participant, please go back and update data entry as applicable.
The Medpace Patient Recruitment and Retention team has officially launched the Vanquish Site Hub. This will serve as a resource to you for study information, updates, and helpful tools for your site.
On the website you will find:
Please don’t hesitate to reach out to your CRA if you have questions!
Please ensure you are not entering a transition date into EDC until the first monthly (Part B) auto injector visit occurs. It is important that this date matches the date of first autoinjector dose. This form is triggering the completing of ePRO diaries and if entered incorrectly will cause issues with collection of the data at correct timepoints. However, we do recommend you complete the IRT transition action from weekly to monthly a week prior to the subjects transition visit.

.png)